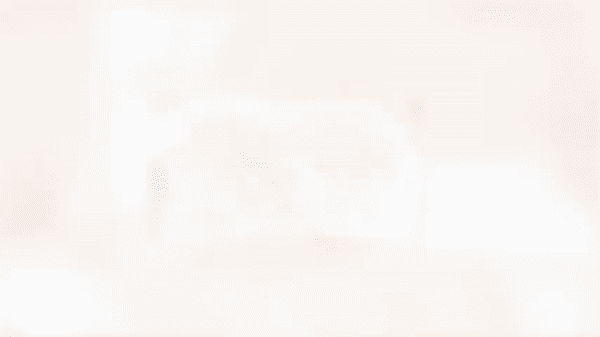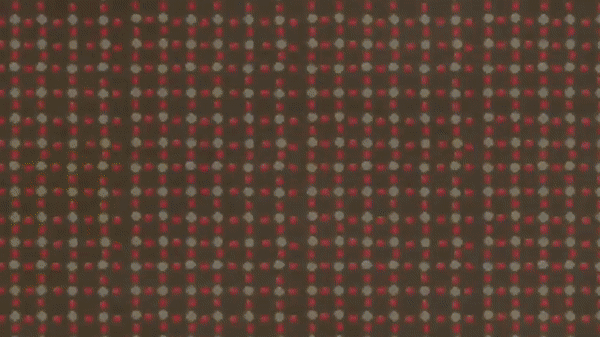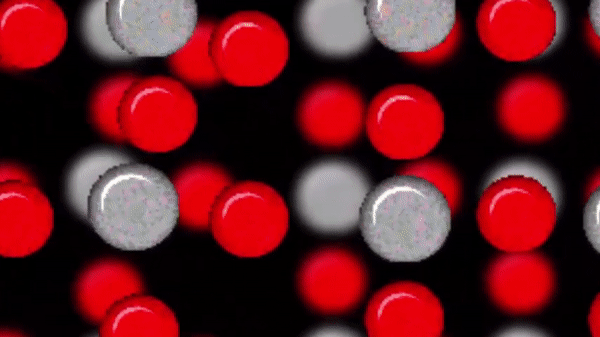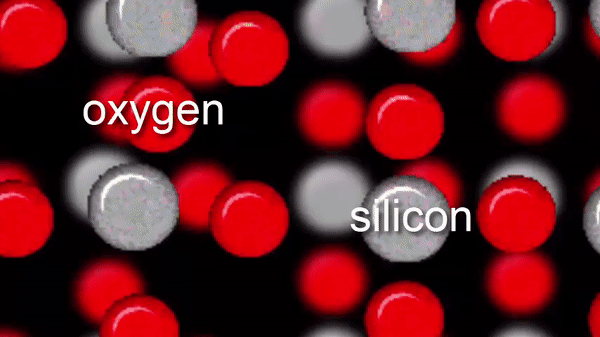tomic School teaches the Big Ideas in science rather than the "baby science" that teachers and students are mandated at the primary school level. Most science curricula worldwide constrain science concepts to the range of students' senses- what they can see and do and touch.
Our core curriculum assumes that primary kids' imaginations are big enough to delve into the microscopic world of the Periodic Table, molecular theory, electric charges and protons, electrons and neutrons. We make it easy and fun for them to fly higher in their science journey. We think that primary school teachers are the best chance our kids have to discover their inner scientist.
A

See our video titles.
Advanced Workshop: Respiration
1st Video: What Are Atoms?
Click on the thumbnail below to see the video.
What They Learn
Your child will discover that their world is build from 92 different kinds of atoms and that they're invisibly tiny.
The zoom at the left simulates how a sand grain is made of silicon and oxygen atoms.
They also learn that LOVE is NOT made of atoms, because it's not matter. To qualify as matter, something has to have mass and take up some space.

2nd Video:
The Size Of An Atom 1
Most kids already know that an atom is too small to see with our eyes. But how small? A thousandth of a metre wide? A millionth? This video answers this.
Starting with a boy under an Oak tree, this video zooms into smaller and smaller objects in a series of steps until finally we can see an atom. Along the way we discover red blood cells, a bacterium, a virus and a sugar molecule. It's quite a ride!
Click on the thumbnail below to see the video.
3rd Video:
The Size Of An Atom 2
Not only do we zoom into an atom, but even tinier sub-atomic particles that make it. Each zooming magnifies our view by ten times, and overall we magnify by ten 24 times. Our smallest object is a neutrino.
4th Video:
What Are Elements? 1
We discover that our whole world is made of only 92 different kinds of atoms, excluding the artificial ones made in the lab. Normally, all these atoms are all mixed up, like in air which has nitrogen, oxgyen and other atoms in it. But sometimes we find or make a substance made of only ONE kind of atom. So we call it an ELEMENT.

5th Video:
What Are Elements? 2
Here we delve into how elements were originally named. After all, our ancestors didn't KNOW that a substance was an element just by looking at it because its atoms were too small to see.
More recently, naming elements has been freed up and now you will find them named after places and even people.
6th Video:
The Periodic Table 1
This has to be the most popular table in the world, and for good reason. It contains ALL the different kinds of atoms in the Universe, including the artificial ones made in the lab- 118 in all.
That's why Atomic School calls the Periodic Table the "Alphabet of the Universe".
Your kids will learn key terms like atomic Symbol and Atomic Number, which help organise the Periodic Table into its special shape.
When you receive this video you will also get a free downloadable A4 Periodic Table.
This is perfect for your fridge and/or bedroom wall. Every child should have a Periodic Table or two.
7th Video:
The Periodic Table 2
Metals and non-metals are the most important categories on the Periodic Table.
Not only are they important as elements in their own right, but they combine to make ionic compounds that are everywhere around us. Think sodium chloride- table salt.
8th Video:
The Periodic Table 3
Hydrogen is special! It defies the main categories and groups on the Periodic Table, and is considered to be a group all by itself.
It is also the lightest element in the Universe which is why they gave it first position on the Periodic Table.
It's also the oldest element in the Universe, a few minutes older than its cousin, helium.


9th Video:
Chemical Bonds 1
Atoms have a habit of sticking onto other atoms to form molecules. These molecules have their own collective personality, that is, their own unique set of properties that are different to the original elements that made them. This makes the world a much more interesting place.
For example, hydrogen and oxygen, both gases, bond to each other to form liquid water molecules, H2O, which is great to drink. It's probably our most important molecule.
10th Video:
Chemical Bonds 2
The bonding videos explain chemical formulas to students. We cover ball-and-stick, molecular formulas and structural formulas because scientists use different forms in different situations. Besides, it's cool to be able to pronounce these advanced terms and show off to your teacher of sibling.
We call H2, H2O, NH3 and CH4 the "Fabulous Four Molecules" and there's a good reason for that. Find out why.
11th Video:
Chemical Bonds 3
Different kinds of atoms can bond to a different number of other atoms. Their bonding power is called its valency.
For example, oxygen can make 2 bonds so it has a valency
of 2. As hydrogen can bond to only 1 other atom, its valency is 1. It follows that 1 oxygen atom can bond with 2 hydrogen atoms, which is why H2O is formed.
These bonding rules are the key to understanding how molecules are made.